Electronic Batch Records
ISA-88 compliant batch execution with digital recipe management, real-time production tracking, and automated regulatory reporting — eliminating paper records and reducing batch review time by up to 80%.
Complete Batch Management
Everything you need for ISA-88 compliant batch execution and electronic records.
ISA-88 Batch Control
Full S88 recipe model with procedures, unit procedures, operations, and phases. Supports modular recipe building and version control.
Digital Recipe Management
Centralised recipe library with version control, approval workflows, and parameter scaling for different batch sizes and equipment.
Electronic Batch Records
Paperless batch documentation with automatic data capture, e-signatures, and audit trails meeting 21 CFR Part 11 compliance.
Real-Time Execution
Live batch progress monitoring with phase tracking, parameter trending, and automatic deviation detection during production runs.
Golden Batch Analysis
Statistical comparison of batch runs against golden batch profiles to identify deviations, optimise yield, and reduce variability.
Regulatory Compliance
Built-in compliance frameworks for FDA 21 CFR Part 11, EU Annex 11, and GAMP 5 with automated audit trail generation.
From Recipe to Release
1. Recipe Configuration
Design batch recipes using the ISA-88 procedural model. Define materials, phases, transitions, and quality parameters with version-controlled master recipes.
- Visual recipe editor with drag-and-drop phases
- Material bill of materials management
- Scalable recipes for different batch sizes
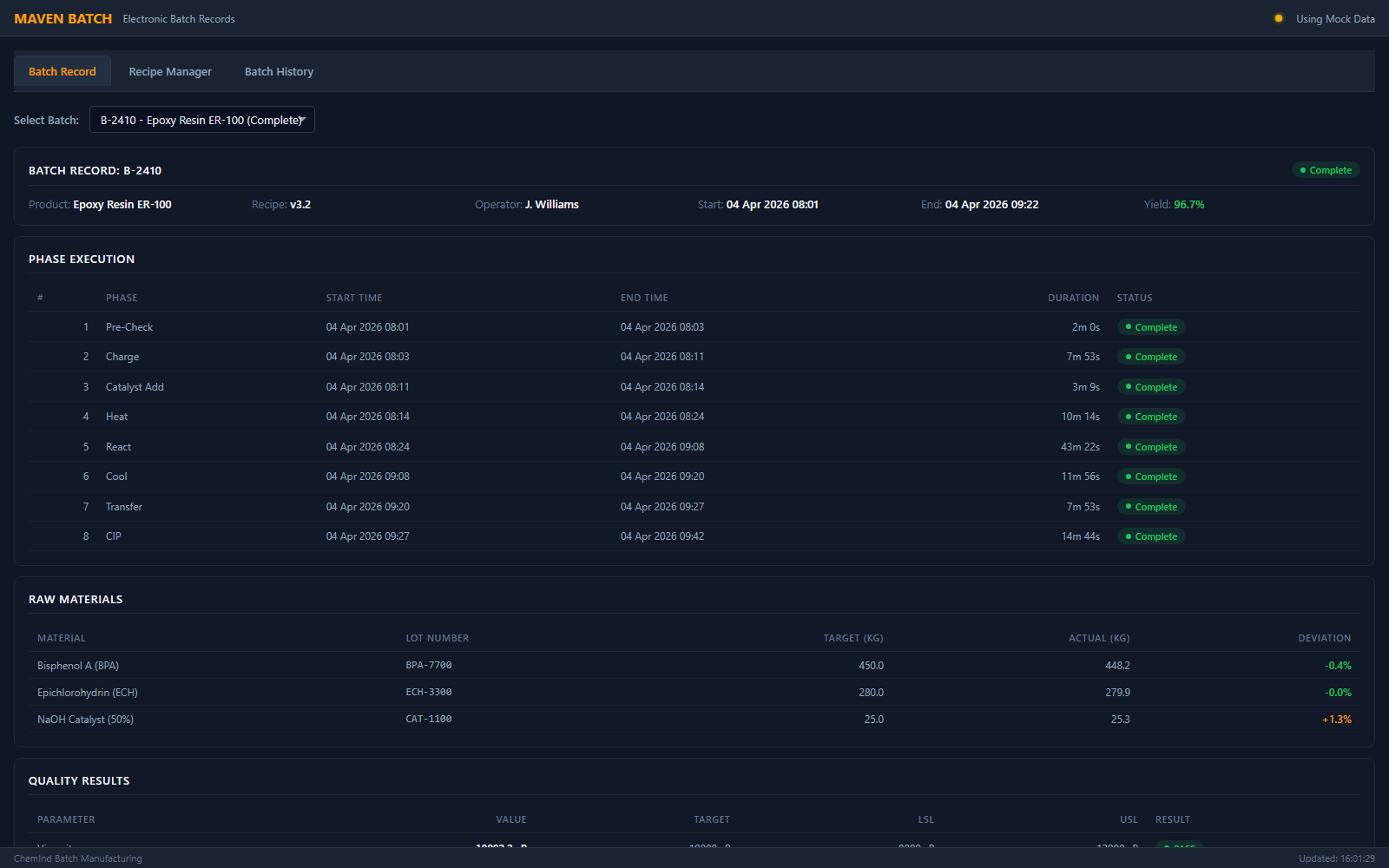
2. Batch Execution
Execute batches with real-time monitoring and automatic data collection. Operators follow guided procedures while the system captures every parameter, event, and deviation.
- Guided operator workflow with e-signatures
- Automatic parameter logging at configurable intervals
- Real-time deviation alerts and hold management

3. Review & Release
Streamline batch review with automated report generation, exception-based review workflows, and electronic approval chains that reduce review-by-exception time from days to hours.
- Automated batch report generation
- Exception-based review highlighting deviations
- Electronic approval chains with audit trail
